Author: Anila B. Elliott, MD - University of Michigan - C.S. Mott Children’s Hospital
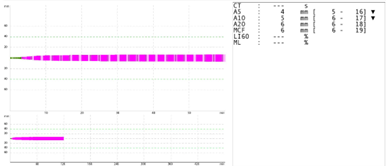
A 2-year-old with congenitally corrected transposition of the great vessels complicated by persistent biventricular dysfunction is undergoing heart transplantation. Following cardiopulmonary bypass (CPB), ROTEM shows normal EXTEM and INTEM. However, the FIBTEM shows the following. The platelet count sent at the same time is measured at 150,000. There continues to be microvascular bleeding from suture lines indicating medical coagulopathy. Administration of which of the following products or factors would be best course of action based on the laboratory findings?
EXPLANATION
Bleeding after CPB in patients with congenital cardiac disease is complex and often multifactorial. Viscoelastic testing, such as rotational thromboelastography (ROTEM), facilitates targeted therapy, reducing the risk of fluid overload, immunologic exposure, and infections1,2. Due to the risk of traditional transfusions, there has been increased interest in the off-label use of factor and fibrinogen concentrates to control bleeding and reduce exposure to donor blood1,3. Data on factor and fibrinogen concentrates in the peri-operative period continues to evolve, but there remain gaps in terms of safety, efficacy, optimal timing and dosing of these novel therapies, especially in the congenital cardiac patient population who have unique hemostatic challenges due to age range, immature hemostatic systems, need for repeat procedures, and sequelae from palliation impacting liver synthetic function1,3.
Primary hemostasis involves platelet plug formation at the site of injury, mediated by von Willebrand factor (vWF). Secondary hemostasis then stabilizes the clot through the coagulation cascade and the conversion of fibrinogen to fibrin, which reinforces the clot4. Recent literature supports targeted fibrinogen replacement for refractory bleeding after CPB when viscoelastic and laboratory testing shows adequate platelet count and clotting factors1,2,4. Fibrinogen deficiency after CPB has been shown to be associated with increased re-exploration rates and blood loss, especially after prolonged bypass times4.
Cryoprecipitate, a component separated from plasma, delivers significant amounts of fibrinogen, factors VIII, XIII, vWF and fibronectin. In clinical practice, its advantages include FDA-approval and broad accessibility; however, the time required to thaw, inconsistent fibrinogen levels, and larger infusion volumes can be significant limitations and there remains a theoretical risk for infectious or immunologic complications5. Fibrinogen concentrates (RiaSTAP and Fibryga) offer more standardized fibrinogen concentration, quick reconstitution, and minimal risk of infection due to purification and viral inactivation. In the US, it is used off-label and can be expensive. Fibryga provides higher factor XIII content than RiaSTAP, which may further support clot stability (Table 1)3.
Although platelet transfusion may be considered for refractory bleeding, in this scenario the normal platelet count, normal EXTEM and INTEM, and abnormal FIBTEM findings suggest fibrinogen deficiency as the likely cause, making fibrinogen supplementation the correct answer (A).
 REFERENCES
REFERENCES
1. Faraoni D, Meier J, New HV, Van der Linden PJ, Hunt BJ. Patient Blood Management for Neonates and Children Undergoing Cardiac Surgery: 2019 NATA Guidelines. J Cardiothorac Vasc Anesth. 2019;33(12):3249-3263. doi:10.1053/j.jvca.2019.03.036
2. Rodriguez, MAP., Jimenez, FM., Ruiz, IF., et al. Evolution of the use of therapeutic fibrinogen concentrate in the massive bleeding guidelines. The Open Anesthesia Journal 2024; 18: http://dx.doi.org/10.2174/0125896458339158240826043207
3. Goodarzi S, Abu-Hanna J, Harper S, Khan D, Morrow G, Curry N. Are all fibrinogen concentrates the same? The effects of two fibrinogen therapies in an afibrinogenemic patient and in a fibrinogen deficient plasma model. A clinical and laboratory case report. Front Med (Lausanne). 2024;11:1391422. Published 2024 May 30. doi:10.3389/fmed.2024.1391422
4. Tirotta, CF., Lagueruela, RG., Madril, D., et al. Use of human fibrinogen concentrate in pediatric cardiac surgery patients. Int J Anesthetic Anesthesiol 2015; 4(2): https://doi.org/10.23937/2377-4630/2/4/1037
5. National Clinical Guideline Center (UK). Blood Transfusion. London: National Institute for Health and Care Excellence (NICE); 2015 Nov. (NICE Guideline, No. 24) 17, Cryoprecipitate: doses. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK338787/
6. Abrahamyan L, Tomlinson G, Callum J, et al. Cost-effectiveness of Fibrinogen Concentrate vs Cryoprecipitate for Treating Acquired Hypofibrinogenemia in Bleeding Adult Cardiac Surgical Patients. JAMA Surg. 2023;158(3):245-253. doi:10.1001/jamasurg.2022.6818
