Authors: Manal Mirreh, MD AND Asif Padiyath, MBBS - Children’s Hospital of Philadelphia
A 13-month-old 8 kg child with hypoplastic left heart syndrome (HLHS) status post Stage 1Norwood followed by bidirectional Glenn at 7 months of age presents for diagnostic cardiac catheterization for hypoxia with oxygen saturation of 60-65%. Superior vena cava (SVC) angiograms demonstrated a diffuse reticular pattern of the pulmonary vasculature, with rapid transit time of contrast into the pulmonary veins, and an absent capillary phase. No decompressing venovenous collaterals were identified on angiography. Indexed pulmonary vascular resistance (PVRi) on FiO2 = 100% and iNO 20ppm was 3.5 WU x m2.
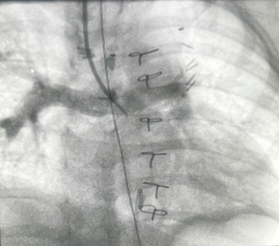
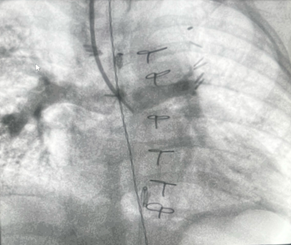
Which of the following surgical interventions is MOST appropriate to improve the systemic oxygen saturation in this patient?
EXPLANATION
The angiographic findings described -a diffuse reticular pattern, rapid contrast transit time from pulmonary artery to pulmonary veins, and absent capillary phase -are pathognomonic for pulmonary arteriovenous malformations (PAVMs), a known complication following superior cavopulmonary anastomosis (Glenn procedure).
PAVMs develop in some Glenn patients due to the absence of hepatic venous blood flow to the lungs. Hepatic venous blood contains an unidentified hepatic factor (HF) that is essential for normal pulmonary vascular development and prevention of PAVM formation1. In the Glenn circulation, pulmonary blood flow comes exclusively from the SVC, which excludes blood from the inferior vena cava (IVC) and hepatic circulation. As a result, hepatic-derived protective factors do not reach the pulmonary vasculature. The absence of these factors leads to abnormal pulmonary angiogenesis, resulting in the development of microscopic and macroscopic PAVMs. Provision of HF in patients with single ventricle physiology and PAVMs has been shown to regress PAVM burden in some patients.2
Patients who remain Glenn-circulation dependent become progressively cyanotic over time. This occurs due to several mechanisms:
1. Somatic growth of the lower body increases inferior vena cava venous return, which is not directed to the pulmonary circulation, while pulmonary blood flow remains fixed and limited to superior vena cava return alone.
2. Development of pathologic veno-venous collaterals that bypass the pulmonary circulation.
3. Formation of pulmonary arteriovenous malformations.
As cyanosis progresses, Glenn patients may require interventions to augment pulmonary blood flow. Strategies include creation of an additional systemic-to-pulmonary shunt, transcatheter closure of veno-venous collaterals, creation of an arteriovenous fistula to increase pulmonary blood flow, or progression to Fontan completion.
Creating an arteriovenous fistula (AVF) (typically brachial artery to vein, and sometimes carotid artery to internal jugular vein) allows IVC blood containing HF to reach the Glenn circulation via the arterial then venous circulations, theoretically promoting PAVM regression while augmenting pulmonary blood flow and improving oxygenation2,3,4. AVF creation is less invasive than either Fontan completion or Glenn takedown, avoiding repeat sternotomy and cardiopulmonary bypass, thereby offering an additional option for single ventricle patients with cyanosis and PAVMs. However, results remain mixed, and reported complication rates vary widely (0-48%); they include limb edema, headaches, and fistula thrombosis. Spearman and colleagues published a retrospective case series including 7 patients with single-ventricle physiology and PAVMs. Six of them survived, and three of them experienced AVF complications (intractable edema and headaches, chest edema, spontaneous occlusion). Improvement in O2 saturation were overall variable and modest.5
Fontan completion (answer choice A) is typically performed at 2-4 years of age. In this 13-month-old 8 kg patient with elevated pulmonary vascular resistance (PVR), this option would carry a high risk of Fontan circulation failure. If the child was older and weighed approximately 12–15 kg, Fontan completion with adequate PVR management would be an appropriate and preferred strategy for definitive management in this scenario. Standard Fontan completion assumes that redirecting hepatic venous blood to both pulmonary arteries will resolve PAVMs through the homogenous distribution of HF. In patients with asymmetric venous anatomy or preferential streaming, a Fontan completion may not guarantee adequate hepatic flow to the affected lung, potentially leaving PAVMs.2
A Glenn takedown to a systemic-to-pulmonary shunt (answer choice B) would restore hepatic blood flow to the lungs but would represent a step backward in the staged single-ventricle palliation pathway and would significantly increase volume load on the single ventricle and therefore may not be the first option to entertain.
Creating an AVF (answer choice C) would allow HF to reach the pulmonary circulation via mixing of IVC blood into the single ventricle, through the systemic arterial then venous systems. 2,3,4. Whilst this strategy yields variable results, it represents a less invasive solution for hypoxemic Glenn patients with AVMs who may not be amenable to Fontan completion in the near future.5
REFERENCES
1. Srivastava D, Preminger T, Lock JE et al. Hepatic venous blood and the development of pulmonary arteriovenous malformations in congenital heart disease. Circulation. 1995 Sep 1;92(5):1217-22. doi: 10.1161/01.cir.92.5.1217. PMID: 7648668.
2. McElhinney DB, Marshall AC, Lang P, Lock JE, Mayer JE Jr. Creation of a brachial arteriovenous fistula for treatment of pulmonary arteriovenous malformations after cavopulmonary anastomosis. Ann Thorac Surg. 2005 Nov;80(5):1604-9. doi: 10.1016/j.athoracsur.2005.05.100. PMID: 16242424.
3. Glenn WW, Fenn JE. Axillary arteriovenous fistula. A means of supplementing blood flow through a cava-pulmonary artery shunt. Circulation. 1972 Nov;46(5):1013-7. doi: 10.1161/01.cir.46.5.1013. PMID: 5081135.
4. Mitchell IM, Goh DW, Abrams LD. Creation of brachial artery-basilic vein fistula. A supplement to the cavopulmonary shunt. J Thorac Cardiovasc Surg. 1989 Aug;98(2):214-6. PMID: 2755154.
5. Spearman AD, Kindel SJ, Woods RK, Ginde S. Arteriovenous fistula creation for hypoxia after single ventricle palliation: A single-institution experience and literature review. Congenital Heart Disease. 2019; 14: 1199–1206. https://doi-org.proxy.library.upenn.edu/10.1111/chd.12828
