Authors: Manal Mirreh, MD AND Deborah A. Romeo, ScM, MD - Children’s Hospital of Philadelphia AND Jeremy Friedman, MD - Cedars-Sinai Medical Center
A 14-year-old with a failing Fontan circulation develops protein-losing enteropathy refractory to medical therapy. Cardiac catheterization is performed to assess candidacy for innominate vein turndown to achieve thoracic duct decompression. Which of the following hemodynamic parameter is BEST predictor of a successful outcome?
EXPLANATION
In the Fontan circulation, systemic venous return flows passively into the pulmonary arteries without a subpulmonic ventricle, leading to chronically elevated central venous pressure (CVP)/Fontan pressure. In patients with the Fontan circulation, the thoracic duct drains into the Fontan system rather than into a lower pressure atrium as in a biventricular circulation. Sustained elevated Fontan pressures increase hepatic lymphatic production while simultaneously obstructing forward lymphatic drainage from the thoracic duct. This combination produces lymphatic hypertension, thoracic duct dilation, and formation of extensive collateral lymphatic channels. As a result, Fontan patients may develop post-operative chylothorax, plastic bronchitis (PB), protein-losing enteropathy (PLE), and ascites as manifestations of lymphatic failure1.
Although lymphatic embolization has proven effective for isolated manifestations, it remains insufficient when multiple compartments are involved simultaneously. An alternative strategy addresses the problem by decreasing thoracic duct (TD) afterload allowing more physiological lymphatic drainage2.
Thoracic duct decompression (TDD) works by redirecting lymphatic flow from a high-pressure venous system (the Fontan circuit) into the lower-pressure atrial chamber. For this to be beneficial, there must be a meaningful pressure differential between where the thoracic duct currently drains and where it will drain after the procedure. TDD, whether performed percutaneously or surgically, remains a highly specialized intervention available at only a select number of centers. Alaeddine et al. from Phoenix Children's Hospital recently reported their early experience with surgical TDD in the single ventricle population (2025), representing one of the first published series describing a systematic surgical approach in this high-risk cohort
Patient selection was limited to those with protein-losing enteropathy (PLE) and preserved single ventricle systolic function. Notably, there were no anatomic exclusions, as the procedure was tailored to each patient's individual anatomy. From a hemodynamic standpoint, the authors carefully reviewed filling pressures and offered the procedure only to patients with a relatively normal single ventricular end-diastolic pressure (≤13 mm Hg). This threshold was deliberately chosen to ensure an adequate pressure gradient existed between the thoracic duct and the recipient atrial chamber, a necessary condition for lymphatic flow to be effectively redirected away from the high-pressure Fontan circulation toward a lower-pressure chamber.3
It may appear counterintuitive, but elevated Fontan pressures and transpulmonary gradient do not preclude TDD. These abnormalities are central to the pathophysiology of Fontan failure and protein-losing enteropathy, reflecting impaired lymphatic drainage rather than a contraindication to intervention. In fact, published transcatheter TDD series from the Children’s Hospital of Philadelphia (CHOP) report median Fontan pressures of 18 mm Hg and transpulmonary gradients of 11 mm Hg, underscoring that patients are often hemodynamically abnormal at baseline.2
A similar experience has been reported by cardiac interventionalists at CHOP, who demonstrated that favorable outcomes following transcatheter TDD were likewise dependent on adequate atrial decompressive capacity, reinforcing that preserved ventricular end-diastolic pressure is a critical determinant of success regardless of whether the approach is surgical or catheter-based.2,3.
Figure 1 is a graphical representation of transcatheter thoracic duct decompression2.
Using fluoroscopic and intravascular ultrasound guidance, a communication is created between the TD (via the innominate vein) and the pulmonary venous atrium. A covered stent is then deployed to maintain this connection and ensure durable patency.
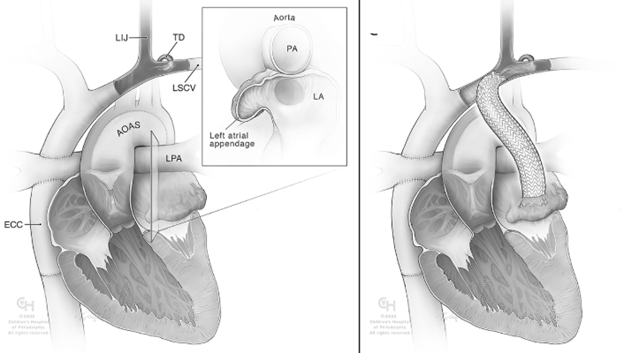
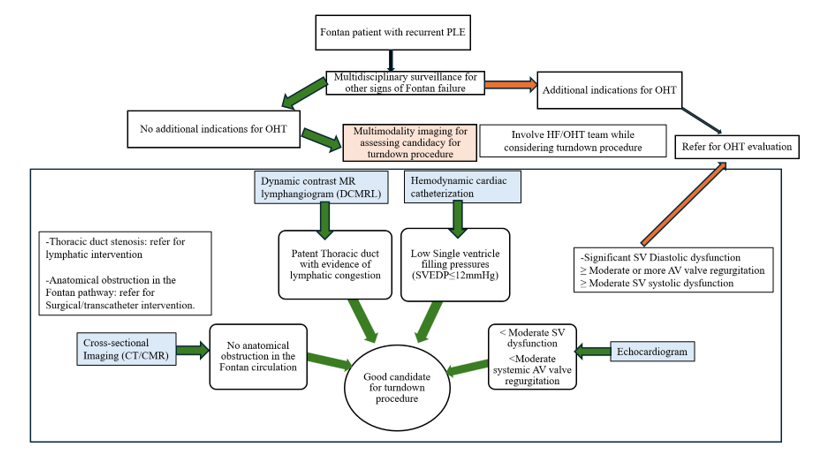
Figure 2 below shows a practical algorithm for patient selection for turndown procedure in isolated PLE.4
REFERENCES
1.Biko DM, DeWitt AG, Pinto EM et al., MRI evaluation of lymphatic abnormalities in the neck and thorax after fontan surgery: relationship with outcome. Radiology. 2019;291:774–780. doi: 10.1148/radiol.2019180877
2. Smith CL, Dori Y, O'Byrne ML, Glatz AC, Gillespie MJ, Rome JJ. Transcatheter Thoracic Duct Decompression for Multicompartment Lymphatic Failure After Fontan Palliation. Circ Cardiovasc Interv. 2022 Jul;15(7):e011733. doi: 10.1161/CIRCINTERVENTIONS.121.011733. Epub 2022 Jun 16. PMID: 35708032.
3. Alaeddine M, Bhat DP, Pohlman J et al. Surgical thoracic duct decompression: The right choice for Fontan-associated protein-losing enteropathy? JTCVS Tech. 2025 May 3;31:133-141. doi: 10.1016/j.xjtc.2025.04.013. PMID: 40641787; PMCID: PMC12237852.
4. Bhat DP, Pohlman J, Almasarweh SI, Gooty VD, Velez D, Alaeddine M. Surgical systemic vein turndown for thoracic duct decompression in Fontan patients with protein-losing enteropathy: role of multimodality imaging. ACC Latest in Cardiology. January 14, 2026.
